|
5/2/2023 0 Comments Serial cloner xdna pet28a I took five well separated colonies and then grew a 5 ml overnight culture. In the following day of transformation, I found many colonies (see attached PPT for picture). I incubated the Gibson reaction at 50oC for 1 hr in a PCR machine and then transformed 2 ul of assembly reaction in 50 ul of NEB 10-beta cell (High efficiency) following the transformation protocol for NEB beta cell as specified by NEB. After that, I set up a Gibson assembly following the instruction of NEB Gibson manual. Then, I digested 8 ul (296 ng/ul) of purified PCR product with DpnI (1 ul) in a 10 ul of total reaction volume (and incubated at 37oC for 30 mins) as suggested by NEB Gibson manual and heat inactivate the DpnI at 80oC for 20 mins. I eluted in 30 ul of nulcease free H2O and then measured the concentration of PCR product using NanoDrop and the concentration was 296 ng/ul. After that, I purified the PCR product using Promega PCR and gel purification kit. After PCR amplification, I checked 5 ul of my PCR product in the gel and found a nice clean single band and the size of this band matches to the expected size (for details see the attached PPT slide). I prepared my vector by PCR (primer designed by NEB builder software). I am using pGex-6p-1 vector to assemble these gBlocks using Gibson assembly. I have designed four gBlocks (each of which is ~1.7 kb) and synthesized them from IDT. I am trying to generate a bacterial operon construct. Could it be my competent cells introducing the mutations? Any help would be greatly appreciated! (Side note: my last two orders of master mix from NEB have smelled bad- almost sulfuric? Did I just not notice this before or is this a sign the mix is tainted?) The positive control still works and gives many colonies. I thought at first the polymerase in the master mix had gone bad, but I have tried buying fresh NEB Gibson Master Mix, and the same kind of mutations still happen. The mutations are often, but not always, near a "seam" where two fragments meet. 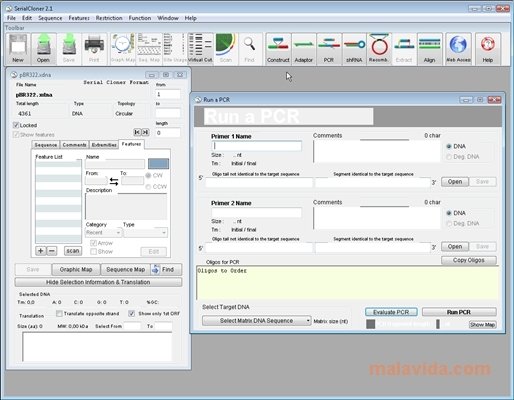
It seems that these mutations are happening either during the assembly or the transformation. 
I got all of my fragments sequenced and I know that before the assembly, they have really clean reads. Most of them look good by restriction digest, but the sequencing results now always show random point mutations that differ from colony to colony, with some having only one mutation, others 5+. 
I am trying to assemble three fragments (900bp, 780bp, 300bp) and a backbone (9.5kb), and I do still get colonies. I have had a lot of success with Gibson reactions in the past, but all of a sudden, not only has my number of total colonies fallen, the percent of correct colonies has also fallen. GGGGGGGCATCGTTTTGTTCGCCCAGGACTCTAGCTATAGTTCTAGTGGTTGGCTACGTATACTCCGGAATATTAATAGATCATGGAGATAATTAAAATGATAACCATCTCGCAAATAAATAAGTATTTTACTGTTTTCGTAACAGTTTTGTAATAAAAAAACCTATAAATATTCCGGATTATTCATACCGTCCCACCATCGGGCGCGGATCCCGGTCCGTTCGAACCCACCATCGGGCGCGGATCCCGTCCGTTCGAACCAGAACCATCGGGCGCGGATCCCGGTCCGTTCGAACCAGAACCACCATCGGGCGCGGATCCCGGTCCGTTCGAACCAGCCACCATCGGGCGCGGATCCCGGTCCGTTCGAGAAGTAATAGTTAATAGATAATAATAGCTCGAGGCAGCGGTACCAAGGTTAATAGATAATAATAGCTCGAGGCATGCGGTACCAAGTTAATAGATAATAATAGCTCGAGGCATGCGGTACCAAGTTAATAGATAATAATAGCTCGAGGCATGCGGTACCAAGCTTGTCGAGAAGTACTAGAGGATCATAATCAGCCATACCACATTTGTAGAGGTTTTACTTGCTTTAAAAAACCTCCCACACCTCCCCCTGAACCTGAAACATAAAATGAATGCAATTGTTGTTGTTAACTTGTTTATTGCAGCTTATAATGGTTACAAATAAAGCAATAGCATCACAAATTTCACAAATAAAGCATTTTTTTCACTGCATTCTAGTTGTGGTTTGTCCAAACTCATCAATGTATCTTATCATGTCTGGATCTGATCACTGCTTGAGCCTAGGAGATCCGAACCAGATAAGTGAAATCTAGTTCCAAACTATTTTGTCATTTTTAATTTTCGTATTAGCTTACGACGCTACACCCAGTTCCCATCTATTTTGTCACTCTTCCCTAAATAATCCTTAAAAACTCCATTTCCACCCCTCCCAGTTCCCAACTATTTTGTCCGCCCACAGCGGGGCATTTTTCTTCCTGTTATGTTTTTAATCAAACATCCTGCCAACTCCATGTGACAAACCGTCATCTTCGCTACTTTTTCCTCTGTCACAGGATGAAAATTTTTCTGTCATCTCTCGTATAATGTTGTATGACTGAATATCACGCTTATTTGCAGCCTGATGGCGATGGACGCCGCCCTGTAGCGGGCGCCATAACCCCGCCGG 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
